PSMA-TRACTr (JANX014)
In April 2026, Janux announced it dosed its first patient in a Phase 1 clinical trial in late-line mCRPC.
JANX014 is a novel double-masked Tumor Activated T Cell Engager (TRACTr) designed to target prostate-specific membrane antigen (PSMA), a protein expressed in prostate cancer tumors and the tumor vasculature. JANX014 is being evaluated in a Phase 1 clinical trial in patients with metastatic castration-resistant prostate cancer (mCRPC) and represents an exploratory extension of Janux’s PSMA TRACTr portfolio.
Prostate cancer is the most common cancer in men and the second most common cancer overall in the United States. PSMA is highly expressed in prostate cancer, supporting the development of PSMA-targeted biologics, including T cell engagers. Clinical experience with PSMA-directed T cell engagers has demonstrated the potential of this approach. JANX014 was designed to build on these insights and to evaluate how additional masking may further refine immune activation, safety, and clinical utility.
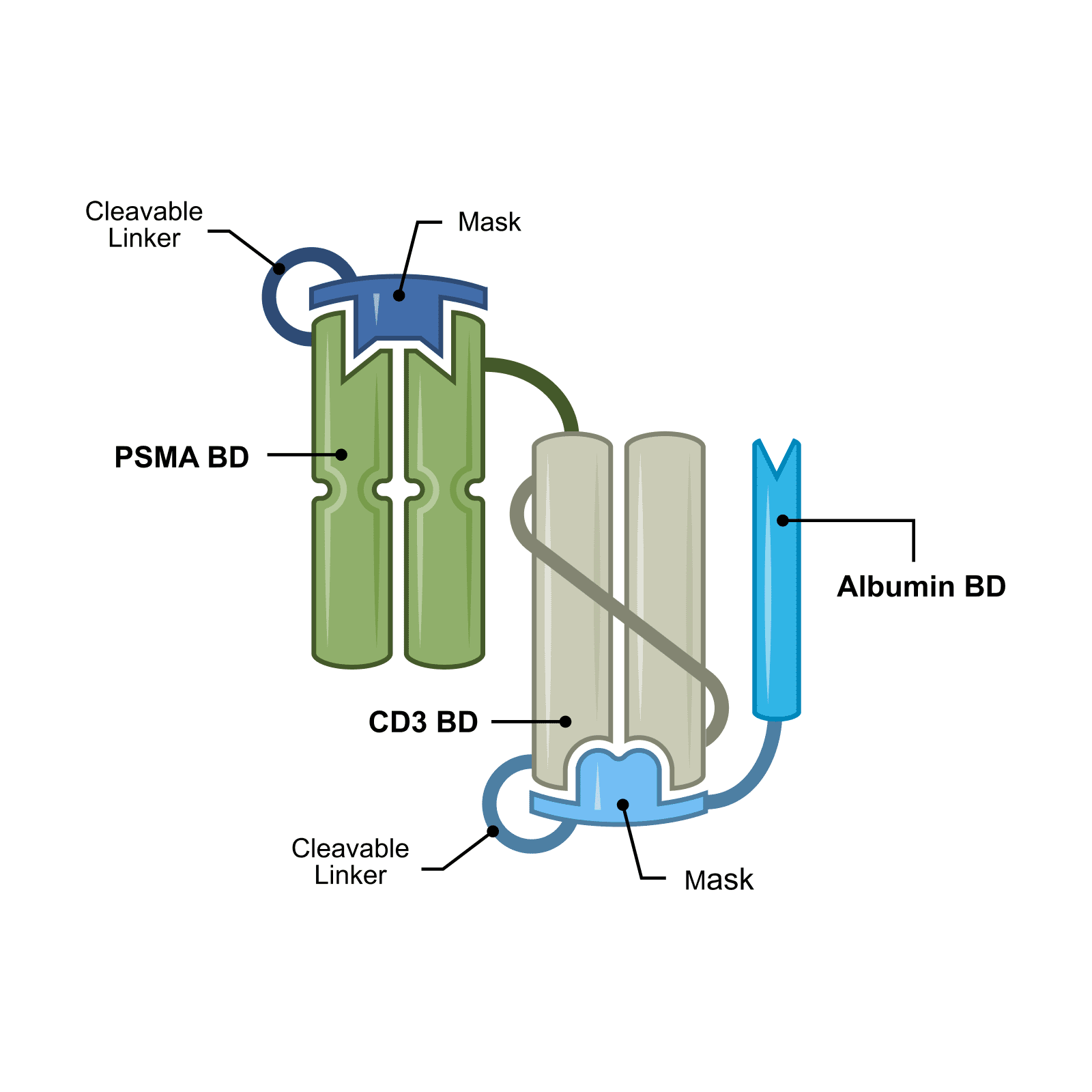
The program builds on clinical experience from JANX007, which has demonstrated meaningful anti-tumor activity and a favorable safety profile, and reflects Janux’s strategy of advancing multiple approaches within its PSMA-targeted portfolio.