PSMA-TRACTr (JANX007)
In December 2025, Janux announced updated interim data for JANX007.
JANX007 is Janux’s lead novel Tumor Activated T Cell Engager (TRACTr). JANX007 is designed to target PSMA, a protein expressed in prostate cancer tumors and the vasculature of tumors and is in the clinic for the treatment of metastatic castration-resistant prostate cancer (mCRPC).
Prostate cancer is the most common type of cancer in men and the second most common type of cancer overall in the United States. PSMA is known to be highly expressed in prostate cancer which has led to the development of PSMA-targeted biologics, including T cell engagers (TCEs). A third-party clinical trial with a continuously infused PSMA-TCE (no longer in development) demonstrated clinical benefit, suggesting the potential of a PSMA-TCE approach.
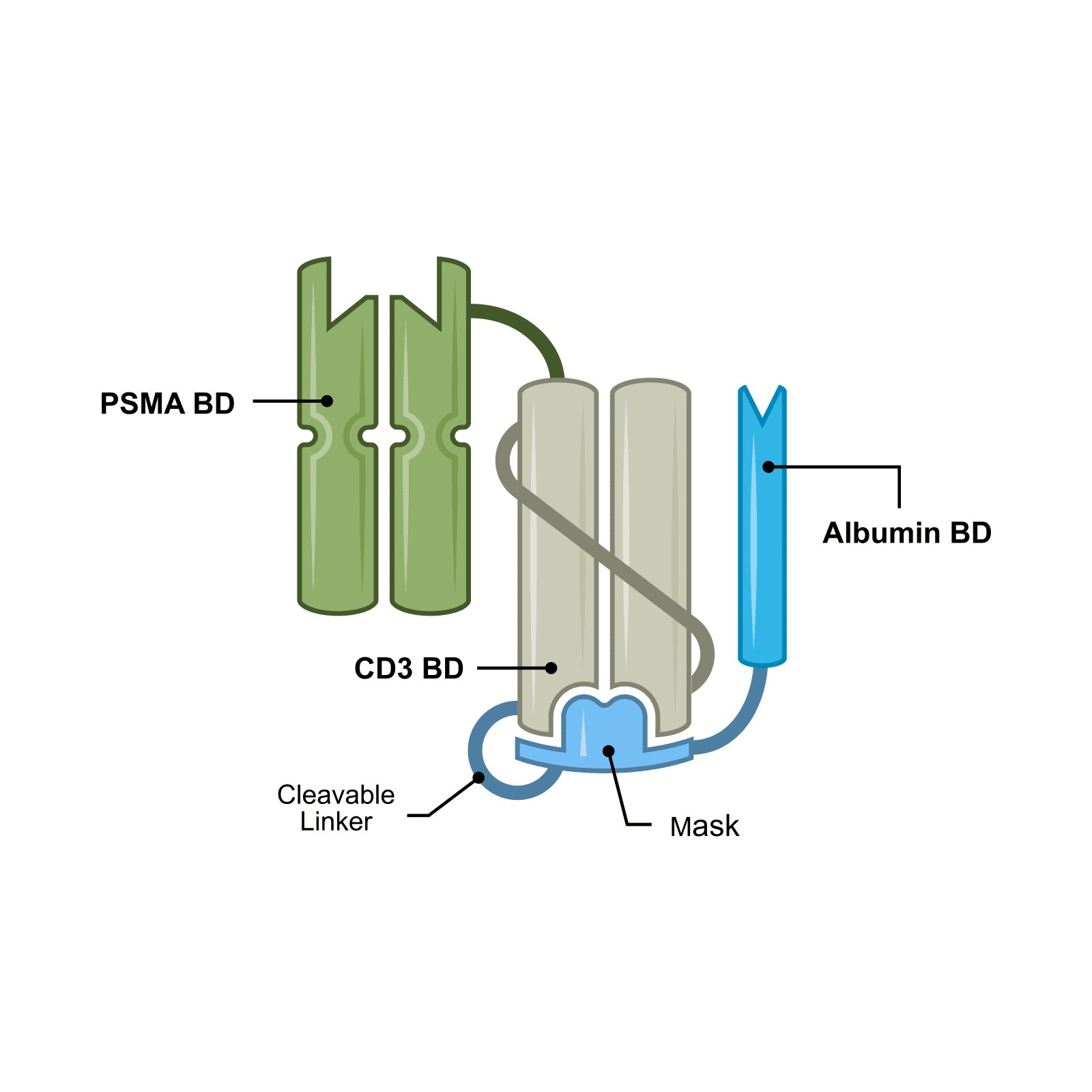
Clinical data from JANX007 have demonstrated substantial anti-tumor activity and a favorable safety profile in patients with mCRPC. The program is advancing with optimized dosing and is being developed in taxane-naïve mCRPC, with additional combination strategies under evaluation.